
Cytokine analysis - ELISA / CBA
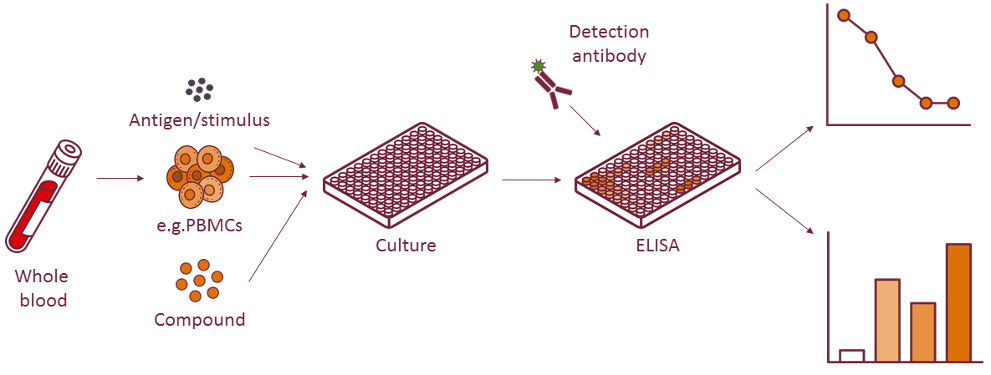
Cytokine production can be measured with different methods. With a cytokine Enzyme-Linked Immuno Sorbent Assay (ELISA), the concentration of ex vivo cytokine levels in serum and plasma, induced cytokine production in our whole blood assays or cytokines produced in culture medium is measured.
This method can be used to screen for the immunogenicity and immunomodulatory properties of compounds and biologicals with regards to innate inflammatory responses, antigen-specific responses, CD4+ T cell skewing and many more immune reactions.
 Advantages of cytokine production analysis by ELISA
Advantages of cytokine production analysis by ELISA
- Cost-effective
- High-throughput
- Monitoring of cumulative cytokine production throughout the culture period
Performing ELISAs is an efficient method to test many samples in different dilutions in one experiment. If, however, you are more interested in measuring various cytokines simultaneously in a small sample volume, the Cytometric Bead Array (CBA) may be more suitable. The CBA is flow cytometry-based, which conveniently allows multiplexing. Alternatively, we also offer secreted protein detection via Luminex for detection of up to 500 different proteins.
Example of cytokine secretion detection by ELISA
Effect of differently induced tolerogenic dendritic cells (DCs) on CD4+ T cell activation
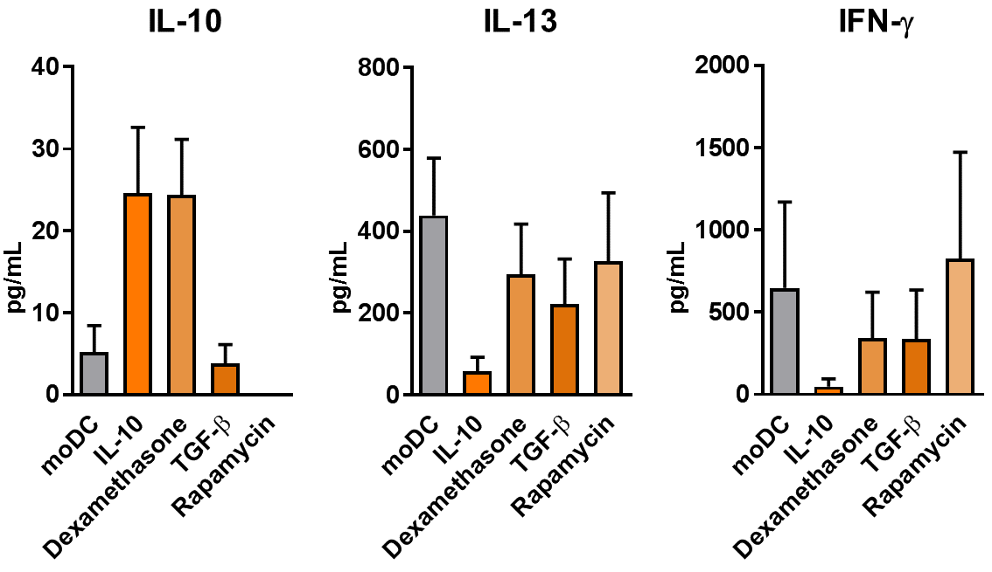 Different types of tolerogenic DCs (matured with indicated compounds) and monocyte-derived DCs (moDCs) were cultured at a 1:10 ratio with allogeneic naive CD4+ T cells. IL-10, IL-13 and IFN-γ production was determined by ELISA of the culture supernatant at day 6. The cultures were performed in duplicate and mean ± SEM of 5 independent experiments is shown.
Different types of tolerogenic DCs (matured with indicated compounds) and monocyte-derived DCs (moDCs) were cultured at a 1:10 ratio with allogeneic naive CD4+ T cells. IL-10, IL-13 and IFN-γ production was determined by ELISA of the culture supernatant at day 6. The cultures were performed in duplicate and mean ± SEM of 5 independent experiments is shown.
Assessing the effect of a compound on a broad panel of cytokines produced by immune cells via a whole blood assay
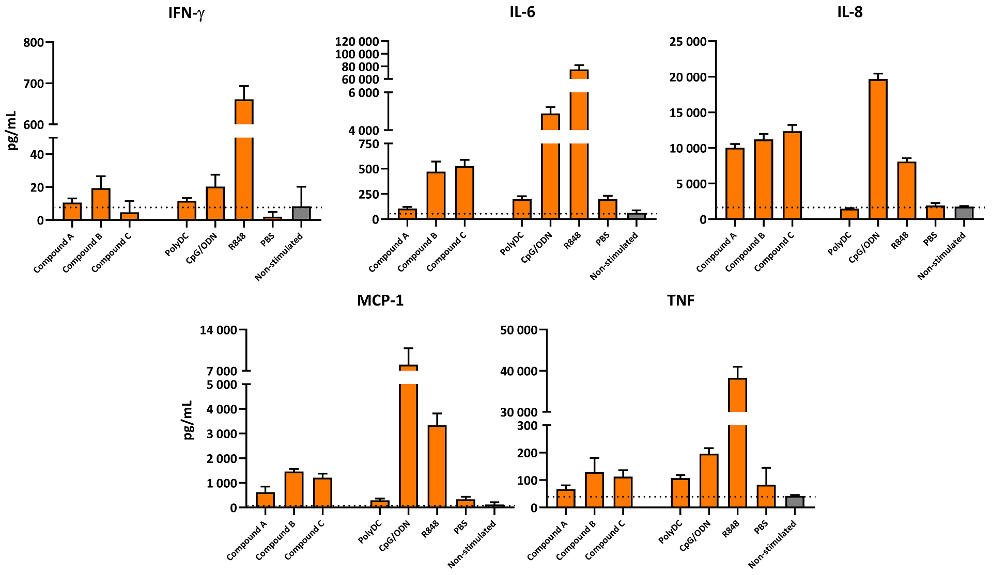 PBMCs isolated from healthy donor whole blood (n=3) was cultured together with potentially immunostimulatory compounds for 6 hours. Serum was collected and cytokine production was assessed by CBA. Graphs show triplicate results from 1 representative donor.
PBMCs isolated from healthy donor whole blood (n=3) was cultured together with potentially immunostimulatory compounds for 6 hours. Serum was collected and cytokine production was assessed by CBA. Graphs show triplicate results from 1 representative donor.
Reference
IL-10-generated tolerogenic dendritic cells are optimal for functional regulatory T cell induction-a comparative study of human clinical-applicable DC
Boks MA, Kager-Groenland JR, Haasjes MS, Zwaginga JJ, van Ham SM, ten Brinke A
Clin Immunol. 2012 Mar;142(3):332-42. doi: 10.1016/j.clim.2011.11.011.
Assay options
We offer cytokine analysis via ELISA in combination with several different standardized stimulation assays, but feel free to contact us for an assay customized to your needs.
Other cytokine analyses
We also offer other methods of cytokine analysis.

