
Healthcare professional experience
Measuring biologics in capillary dried spot samples
To enhance implementation of TDM of biologics in routine care we started developing a self-sampling method for biologic measurements. First, we compared adalimumab and anti-adalimumab antibody (ADA) concentration measurements in dried blood spots (DBS) obtained from finger prick with measurements in serum obtained via venipuncture, from patients with rheumatic inflammatory diseases. Adalimumab and ADA concentrations obtained from the finger prick/DBS method correlated well with serum results from the same patient (correlation coefficient > 0.87). Follow-up studies confirmed this finding, as well as validating measurement of infliximab in DBS.
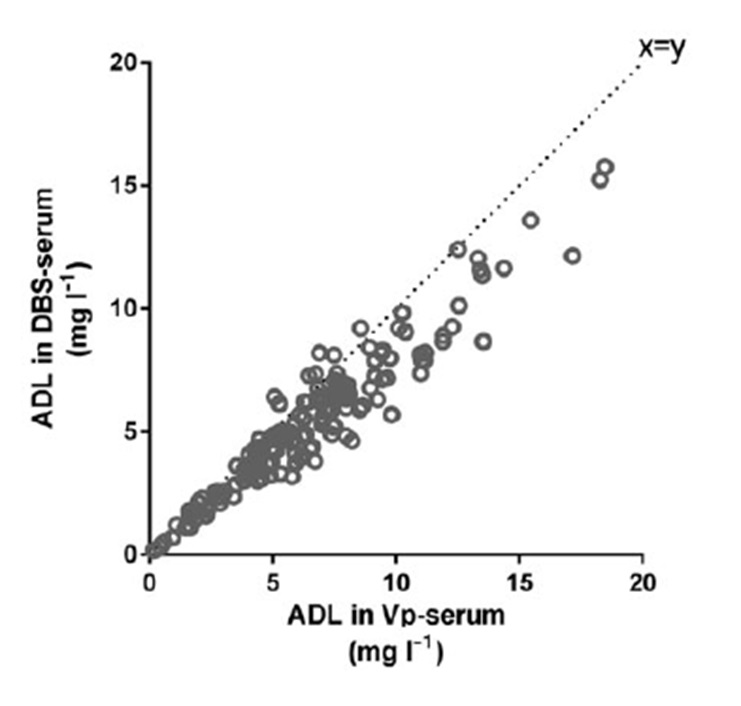
Figure 1: Corrrelation between adalimumab level in venous serum versus capillary serum from fingerpricks.
Measuring biologics in capillary wet blood samples
We extended our validation to comparing the use of finger pricks to measure concentration of monoclonal anti-inflammatory biologics infliximab, natalizumab and vedolizumab in wet capillary blood samples. In one study capillary blood samples by finger prick and intravenous blood samples were collected to assess drug concentrations prior to intravenous administration of the biologics infliximab (n=46) and vedolizumab (n=35) (Otten et al., (2023)). The finger prick was performed by the patient and capillary blood was sent by postal service to us for analysis. Serum concentration of the biologics were analysed with our in-house developed enzyme-linked immunosorbent assay and results were compared using Blant-Altman and Spearman correlation. Serum infliximab and vedolizumab concentrations measured in capillary and intravenous blood samples correlated well (r = 0.99, P < 0.001) and showed a mean bias of 0.42 ug/ml and 0.72 ug/ml, respectively, demonstrating excellent agreement. This study was conducted in collaboration with the Universitair Medisch Centrum Groningen and supported by clinicians. Please find more validation studies on biologics here.
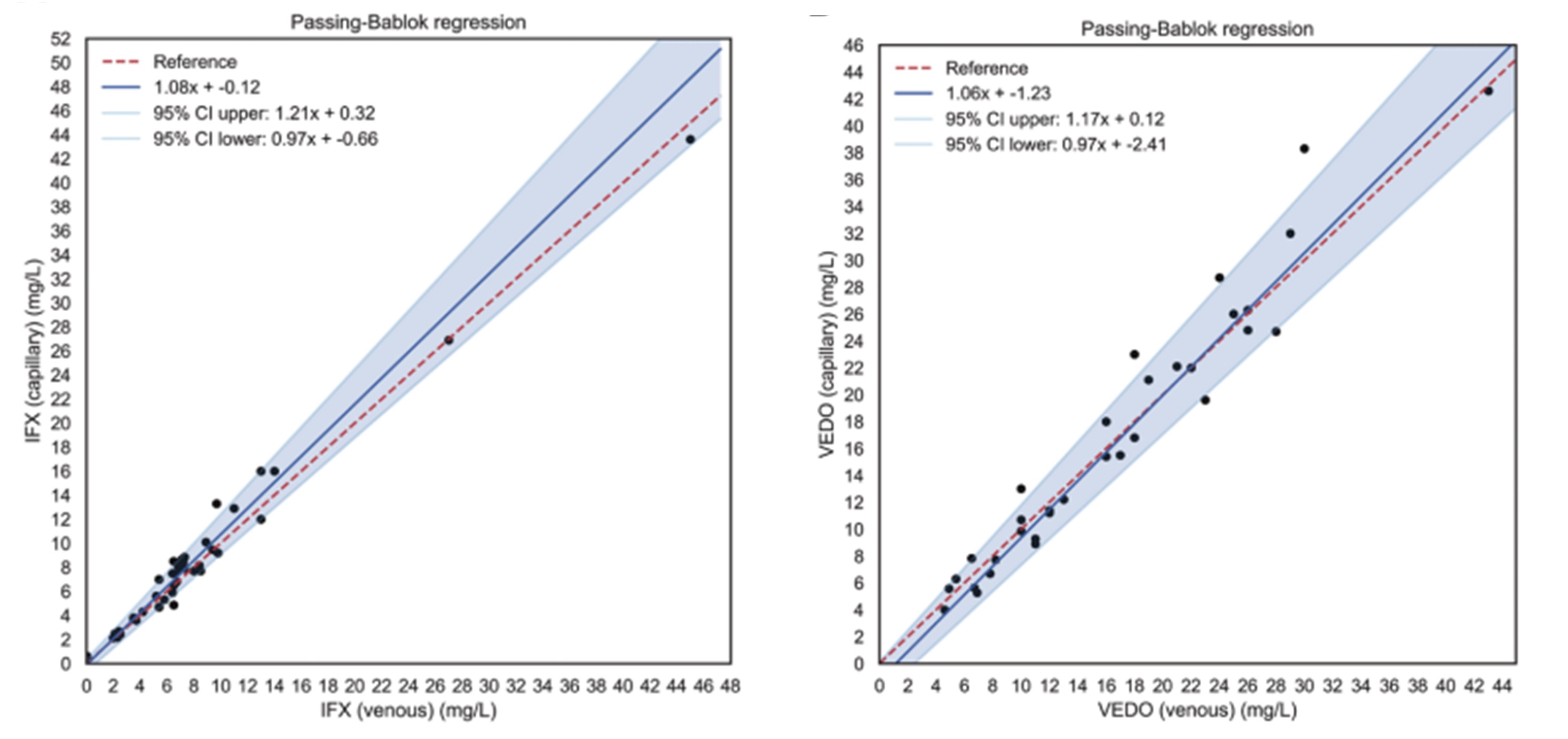
Figure 2. Corrrelation between infliximab level (left panel) and vedolizumab (right panel) in venous serum versus capillary serum from fingerpricks.
Testing COVID-19 antibodies in 100,000 people
During the COVID-19 pandemic Sanquin Research & Lab services (Dr. Theo Rispens, principal investigator Immunopathology) together with Sanquin Diagnostic Services utilised their already developed tests for antibody measurements in capillary blood and validated SARS-CoV-2 serology assays to monitor SARS-CoV-2 antibody responses in capillary blood collected by finger pricks (see our published studies).
We learned how to support large cohorts logistically. These studies were performed in collaboration with different consortia (see below). All studies together we have collected capillary blood using finger pricks in almost 100,000 healthy and immune-compromised patients to determine SARS-CoV-2 antibody development after vaccination. This valuable knowledge about vaccination responses in patients taking immunosuppressive medication were also shared preliminary with the Health Counsel of the Netherlands to help make decisions on national booster campaigns. Our studies have also created a unique biobank of longitudinal samples of after covid vaccination, allowing to answer many other scientific questions. These large scale studies were performed in a short period of time, which was not possible without using finger pricks that were send to the participants from a central point.
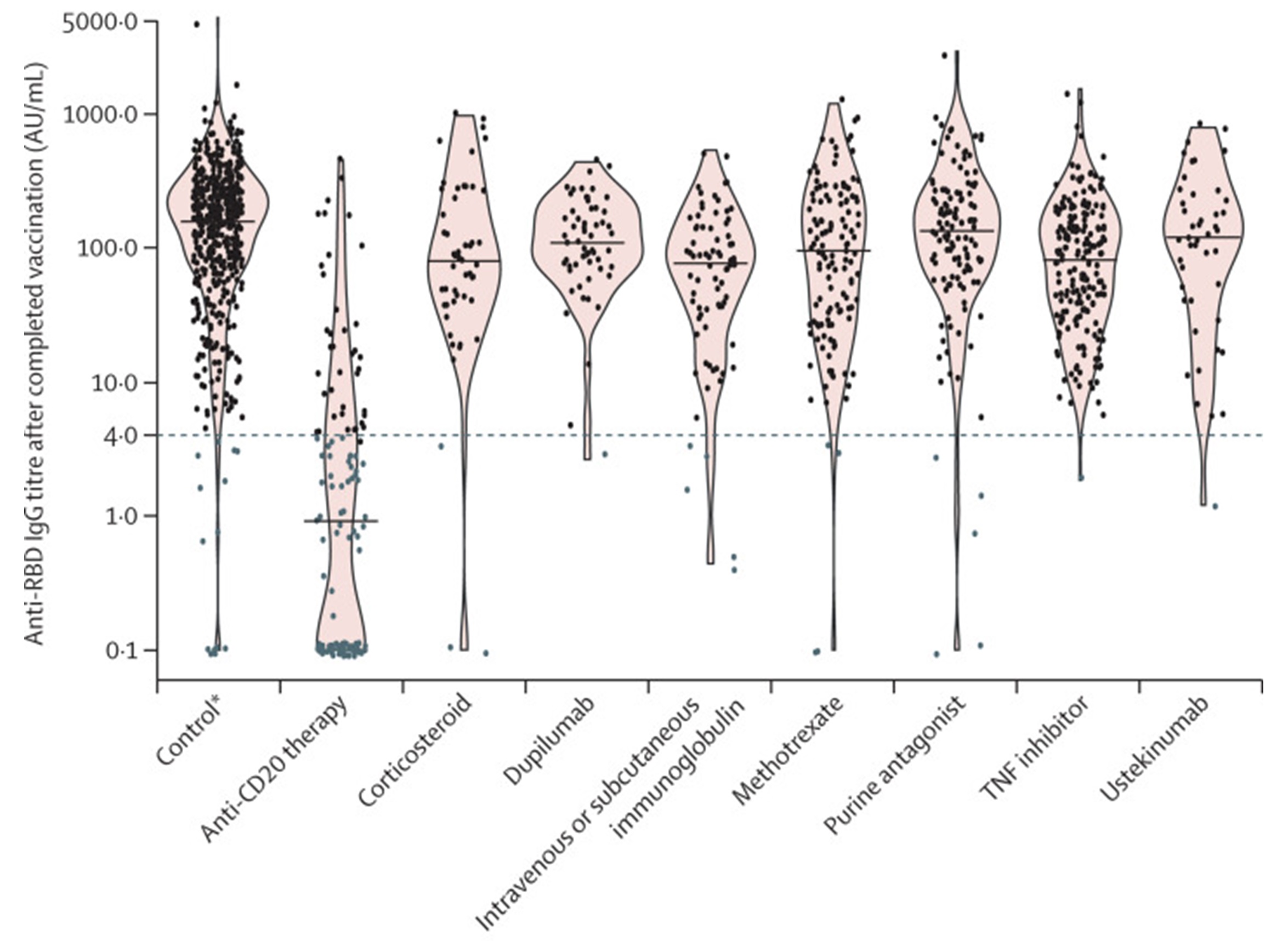
Figure 3. The IgG anti-RBD response of individual patients, using the indicated medication, after their first COVID-19 vaccination. Samples were collected via fingerpricks at home.
Please find a link to the studies below:




